Neonatology
Session: Neonatal Pulmonology - Clinical Science 5: New-Old ideas, Stem Cells
576 - Regulatory Profile of Human Preterm Umbilical Cord Derived Mesenchymal Stem Cells Exposed to COVID-19
Monday, May 6, 2024
9:30 AM - 11:30 AM ET
Poster Number: 576
Publication Number: 576.2987
Publication Number: 576.2987
- SJ
Shruthi Janardhan, MD (she/her/hers)
Fellow, Neonatal-Perinatal Medicine
University of California, Irvine, School of Medicine
Orange, California, United States
Presenting Author(s)
Background: COVID-19 caused by the SARS-CoV-2 virus has led to a worldwide pandemic with oxidative stress, inflammatory cascade, and cytokine storm as main causes of morbidity and mortality. Covid-19 infection in pregnant women varies from asymptomatic to severe disease. Bronchopulmonary dysplasia (BPD) is a chronic debilitating lung disease of preterm infants leading to arrested alveolar development. Oxidant stress along with lung inflammation are leading etiologic factors for BPD. Our published work has shown protection of BPD by mesenchymal stem cells (MSCs) and their exosomes (MSC-exo). This study evaluated biologic efficacy of MSC-exo harvested from umbilical cords of preterm infants with and without Covid-19 infection at delivery.
Objective: We hypothesized that SARS-CoV-2 infection of the pregnant mother at the time of preterm delivery will lead to a decline in biologic efficacy of umbilical cord derived MSC-exosomes. Our aims included generation of MSCs from preterm umbilical cords, assessment of their growth and differential potential, generation of MSC-exo followed by MSC-exo content analysis.
Design/Methods: The study was approved by the Institutional IRB. The umbilical cords at preterm delivery (24-28 weeks’ gestational age) were collected from 6 SARS-CoV-2 positive mothers and 6 SARS-CoV-2 negative mothers (control) after informed consent. The umbilical cord Wharton’s jelly plastic adherent cells harvested, propagated, and immunodepleted to obtain MSCs. MSC duplication and differentiation time assessed between two groups. MSC-exo isolated using our established protocols. MSC-exo proteomics analysis done to determine secreted biomarkers. MSC-exo microRNA (miRNA) levels determined using TaqMan RNA kits per manufacturer protocols.
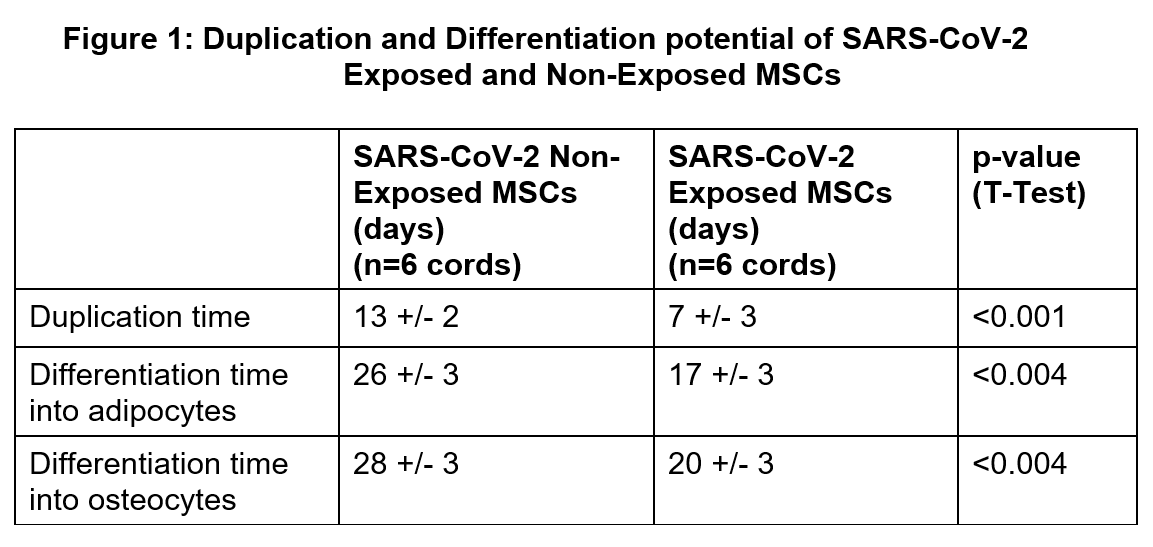
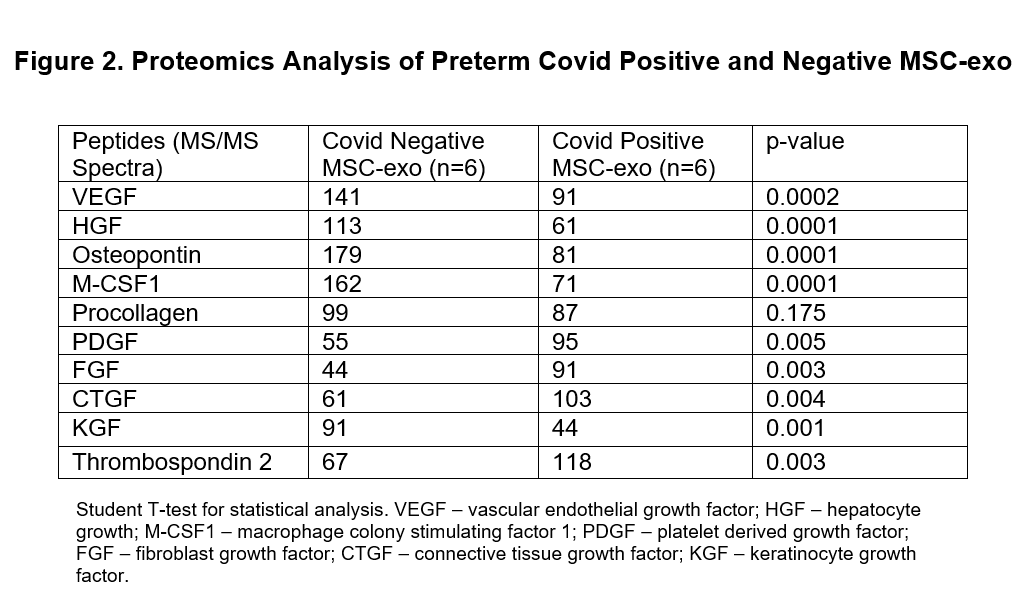
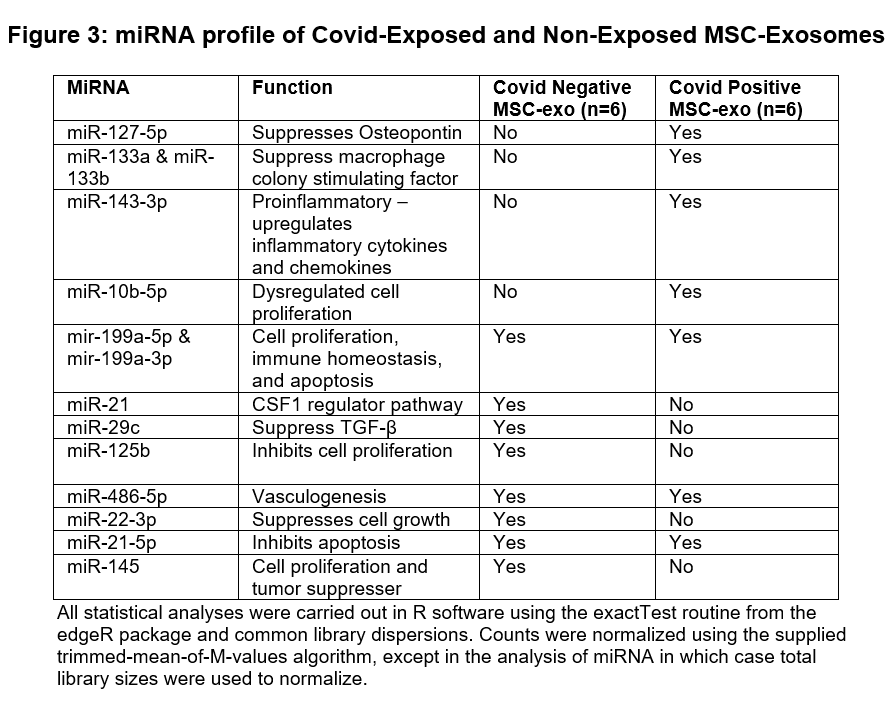
Results: SARS-CoV-2 positive MSCs had much shorter duplication time and differentiation time (adipocytes and osteocytes) when compared with negative controls (Figure 1). Proteomics analysis identified significantly lower lung vascular and epithelial injury preventive biomarkers concentration in SARS-CoV-2 positive MSC-exo compared to control MSC-exo (Figure 2). Similarly, SARS-CoV-2 positive MSC-exo fraction was deficient in lung injury and repair miRNAs when compared with control MSC-exo fraction (Figure 3). All these results were statistically significant.
Conclusion(s): SARS-CoV-2 infection at the time of delivery alters the biologic potential of human preterm umbilical cord derived MSCs with lack of protected biomarkers and miRNAs involved in lung injury and repair.